 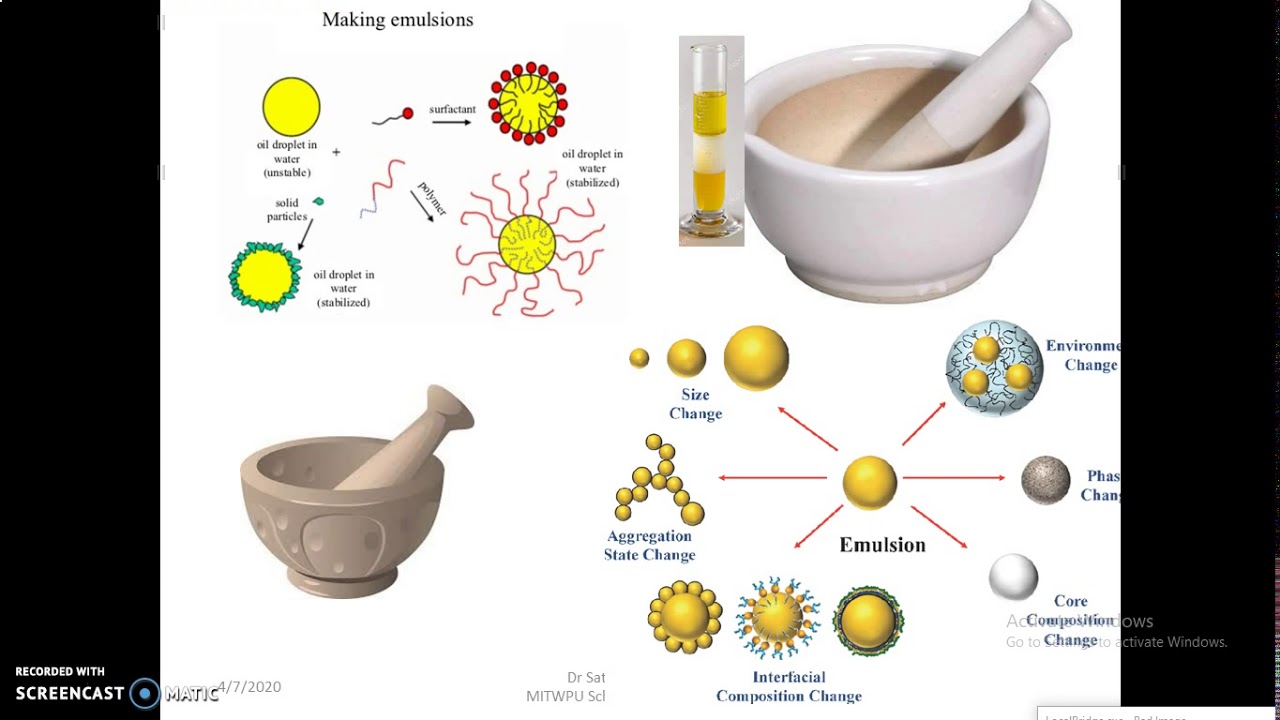
As we know that there are mainly two types of emulsion oil-in-water $(O/W)$ and water -in-oil $(W/O)$.Hence milk is an oil-in-water type emulsion because in case of milk fat dispersed in water. The tactile and spreading properties of emulsions are different from its components. Note : The emulsion has physical properties that differ from its two components. Hence in the given problem option C is correct which Milk is an emulsion in which milk fat is dispersed in water. Milk is an oil in water type emulsion because in milk fat globules are dispersed in water. Casein which is a lyophilic sol which stabilizes the milk emulsion so it acts as a emulsifier for milk. 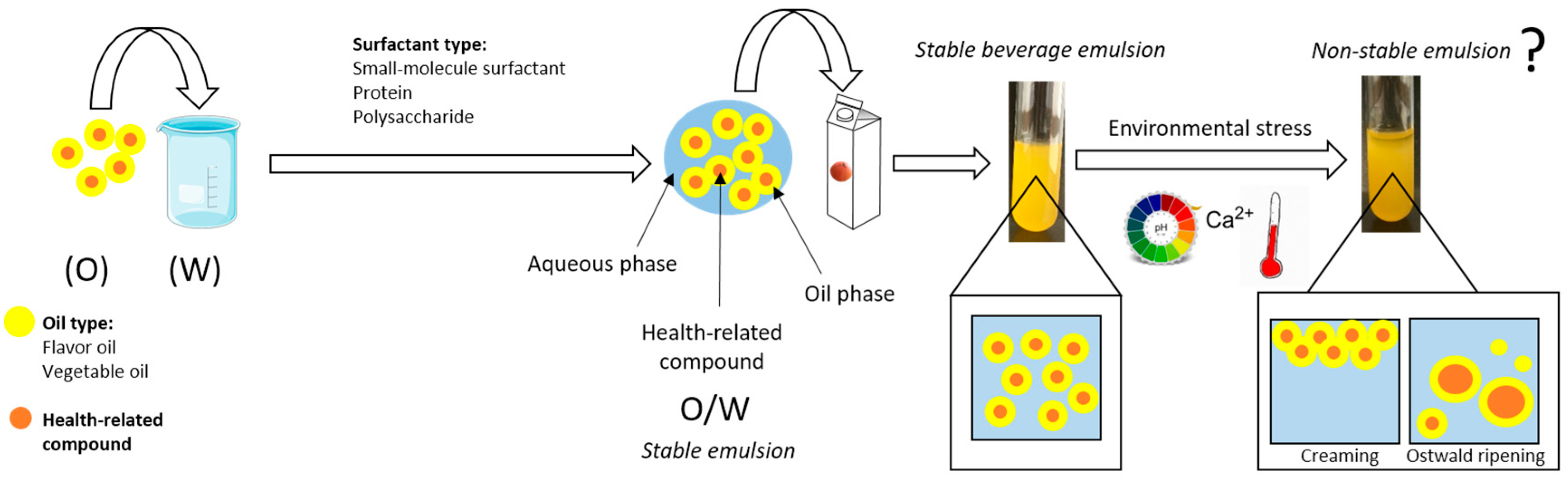
Milk is an emulsion where butterfat globules are suspended in water. > The word emulsion is taken from the Latin words emulgere and emulsum which means milk. It is a colloidal solution where dispersion and dispersed medium both are liquid. Emulsions are used to make medicines more palatable (pleasant taste ).It is also used in skin care products to lock moisture and hydration of skin. > Emulsions are very useful in the pharmaceutical industry. Stable emulsion means it would not be separated with the change in conditions like temperature and time. With the use of an emulsifier stable emulsion can be formed. These are colloids where both phases are liquids. See how many more you can find you will be surprised.Hint : We can define emulsion as the mixture of two liquids and one of the liquids is immiscible liquid which means that the liquids would not mix. Colloids play a significant role in our lives but they are often overlooked or taken for granted. In this experiment we have looked at some very common colloids that can be found and made in our kitchens at home. You probably found that the individual components were quite runny and slid down the ruler quickly, while the colloids you made from them where quite thick and stayed as blobs or slid down slowly – the colloids are more viscous than their components. This will give you an indication of the viscosity of the liquid. Then take each of the individual components and the colloids you have just made and measure the time it takes for a drop of each liquid to travel down the ruler by 10 cm. The fat (butter) in milk is the classic example of an emulsion, drops of one liquid dispersed throughout another. In these two simple experiments, did you notice how the physical properties and appearance of the mixtures changed when the two components were mixed together?Ī good way to scientifically measure the difference in physical properties would be to take a ruler and incline it at an angle of 45 degrees. Keep adding drops of olive oil until you get an emulsion that looks similar to mayonnaise. Using the whisk, mix the two components together. 
You will need two egg yolks (separated from the white), a small amount of olive oil and a whisk.This is an example of a gas-liquid colloid, where gas is trapped within a liquid to form a stable mixture. Take the whisk and whip the cream until it is very thick.You will need a whisk and some double cream.Both of these colloids are found in our everyday lives and will be very familiar to you. Let’s look at a gas-liquid colloid (foam) and a liquid-liquid colloid (emulsion). The table shows a few examples of these combinations Try this out These components can be a mixture of gas-liquid, liquid-liquid, solid-liquid or solid-gas. When separated, the two liquids are often immiscible from one another.Ī colloid is therefore a stable mixture that is made up from two or more components. Milk is a mixture of water and milk fat and mayonnaise is a mixture of oil and water, which is stabilised further by proteins in the egg yolk. Well known emulsions are milk and mayonnaise. An emulsion is a special type of colloid made up from a mixture of two liquids which form a stable substance that has different physical properties to the two individual liquids. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
